Pathological Mechanisms and the Efficacy of Teeth Scaling
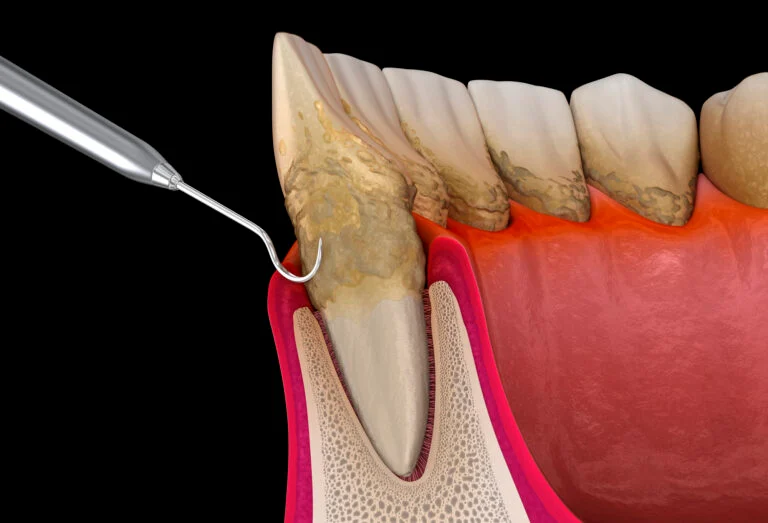
Within the clinical repositories of SmileNote, the analysis of periodontal interventions requires a strict adherence to biological evidence and physiological mechanisms. The procedure clinically designated as teeth scaling is a fundamental therapeutic modality aimed at arresting the progression of periodontal pathogenesis. To understand why teeth scaling is initiated in a clinical setting, one must objectively evaluate the microbiological etiology of dental calculus, the localized host immune response to bacterial biofilm, and the mechanical alteration of the tooth surface. This analysis deconstructs the evidence-based mechanisms through which subgingival and supragingival debridement alters the oral microenvironment and stabilizes the periodontium.
Etiology of Calculus Formation and the Role of Teeth Scaling
The oral cavity represents a highly dynamic ecosystem where the acquired pellicle—a proteinaceous film derived from salivary glycoproteins—coats the enamel surface almost immediately following mechanical cleansing. Within hours, early colonizing bacteria, predominantly gram-positive cocci, adhere to this pellicle. Over subsequent days, a shift in the microbial flora occurs, introducing pathogenic gram-negative anaerobes. If this biofilm is not mechanically disrupted, salivary calcium and phosphate ions precipitate into the organic matrix.
Biofilm Mineralization and Mechanical Disruption
This mineralization process transforms the soft biofilm into a calcified accretion known as dental calculus. Calculus itself is inherently porous and non-pathogenic; however, its rough topography serves as an ideal, retentive reservoir for a constant layer of viable, unmineralized pathogenic biofilm. When addressing the core question of what is teeth scaling, the primary mechanical objective is the complete obliteration of this mineralized lattice. By removing the calculus, the clinician eliminates the retentive surface area that harbors the bacterial load, thereby reducing the localized bio-burden to a level that is manageable by the host’s innate immune system. The American Dental Association acknowledges this mechanical intervention as the critical first step in arresting the microbial succession that leads to attachment loss.

Hemodynamic Response to Periodontal Debridement
The presence of mineralized accretions below the gingival margin induces a chronic inflammatory state within the adjacent soft tissues. This localized immune response is characterized by distinct hemodynamic alterations. The vascular network within the gingival connective tissue undergoes profound vasodilation and increased capillary permeability. This physiological shift is designed to facilitate the rapid migration of polymorphonuclear leukocytes and macrophages to the site of the bacterial challenge.
Vascular Engorgement and Resolution
Clinically, this vascular engorgement manifests as erythema, edema, and a propensity for bleeding upon probing. Following the precise execution of teeth scaling, the removal of the antigenic stimulus triggers a rapid resolution of these hemodynamic changes. The cessation of the bacterial insult allows the capillary beds to constrict and return to their baseline permeability. Consequently, the clinical signs of inflammation subside. The reduction in edema allows the marginal gingiva to adapt closely to the root surface, effectively reducing the depth of the periodontal pocket and re-establishing a physiological biological seal against future microbial ingress.
Immunological Mediators Modulated by Teeth Scaling
The tissue destruction observed in progressive periodontal disease is not primarily caused by bacterial exotoxins, but rather by the host's own hyper-reactive immune response.
Cytokine Cascade and Tissue Degradation
The persistent presence of subgingival calculus triggers the continuous release of pro-inflammatory cytokines, specifically Interleukin-1 beta (IL-1β), Tumor Necrosis Factor-alpha (TNF-α), and Prostaglandin E2 (PGE2). These biochemical mediators stimulate the production of matrix metalloproteinases (MMPs), which degrade the collagenous framework of the periodontal ligament, and activate osteoclasts, which resorb the alveolar bone.
The therapeutic intervention of teeth scaling directly modulates this immunological cascade. By mechanically debriding the root surface of the inciting antigens, the local synthesis of IL-1β and TNF-α is abruptly downregulated. This biochemical shift halts the activation of MMPs and osteoclasts, thereby arresting the degradation of the connective tissue attachment and alveolar bone. Histological analyses of periodontal tissues post-debridement confirm a transition from a leukocyte-infiltrated, inflammatory lesion to a fibroblast-rich environment capable of collagen synthesis and tissue repair.
The clinical efficacy of periodontal debridement is rooted in basic biological sciences. The procedure fundamentally disrupts the mineralization of pathogenic biofilm, reverses localized vascular hyper-permeability, and halts the destructive cytokine cascade responsible for tissue degradation. As an evidence-based intervention, it remains the primary mechanism for restoring homeostasis to the periodontium through professional teeth scaling.