Histological and Osseous Integration Phases: A Chronological Analysis of the Dental Implant Process Timeline
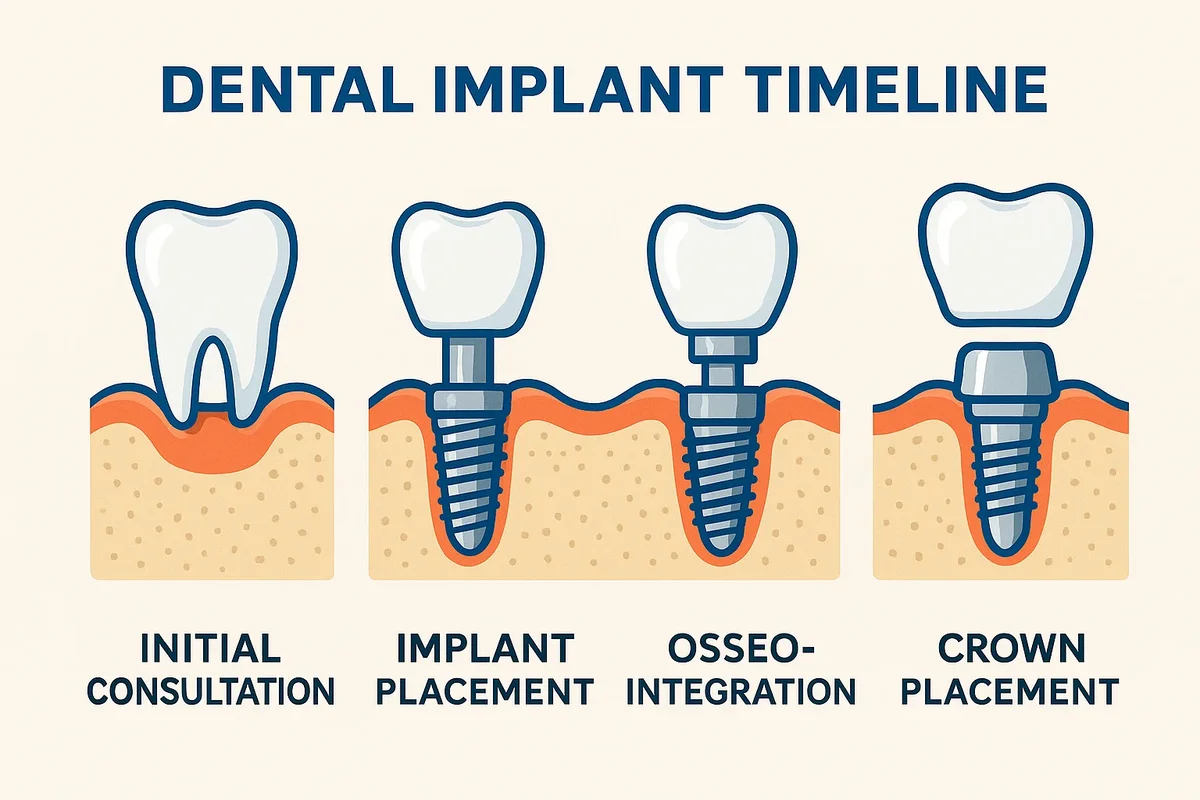
Welcome to SmileNote. In the discipline of implant dentistry, the restoration of edentulous spaces is governed by strict biological parameters. The dental implant process timeline is not an arbitrary schedule but a reflection of the physiological rates of hemostasis, inflammation, proliferation, and remodeling. Understanding the timeframe for dental implants requires a deconstruction of the cellular events occurring at the bone-implant interface (BII). From the initial osteotomy to the final prosthetic loading, every phase is dictated by the host's ability to synthesize woven bone and subsequently remodel it into load-bearing lamellar bone. This analysis examines the biological rationale behind the standard clinical intervals.
Phase I: The Osteotomy and Primary Stability
The initiation of the dental implant process timeline begins with the surgical phase.
The Hemostatic Response
Upon the creation of the osteotomy (surgical drilling), vascular disruption occurs within the alveolar bone. The immediate physiological response is hemostasis. Within seconds, a fibrin clot forms, filling the gap between the titanium surface and the host bone. This clot serves as a provisional matrix for cell migration. The stability of this clot is critical; micromotion exceeding 150 microns during this phase can disrupt the fibrin network, leading to fibrous encapsulation rather than osseointegration. The initial 24 to 48 hours are defined by the release of growth factors such as Platelet-Derived Growth Factor (PDGF) and Transforming Growth Factor-Beta (TGF-β), which recruit neutrophils and macrophages to debride the wound.
Primary Stability Metrics
Clinically, the success of this phase is measured by insertion torque values (ITV) and Implant Stability Quotient (ISQ). High primary stability (ITV > 35 Ncm) is achieved through mechanical friction between the implant threads and the bone trabeculae. However, this mechanical stability is transient. As the body mounts an inflammatory response to the trauma of surgery, osteoclasts begin to resorb the bone immediately adjacent to the implant. This leads to a temporary dip in stability, typically observed between weeks 3 and 4 of the timeframe for dental implants. This biological trough represents the transition from mechanical retention to biological fixation.

Phase II: Proliferation and Woven Bone Formation
The proliferative phase, extending from week 1 to week 6, is characterized by angiogenesis and osteogenesis.
Angiogenesis and Vascularization
New blood vessels sprout from the existing vascular network, penetrating the granulation tissue. This neovascularization provides the oxygen tension required for osteoblast differentiation. Mesenchymal stem cells migrate to the implant surface and differentiate into osteoblasts. These cells secrete a non-collagenous matrix that rapidly mineralizes to form woven bone. Woven bone is highly cellular and disorganized, lacking the structural anisotropy of mature bone. While it bridges the gap, its biomechanical competence is limited. It provides initial biological stability but is susceptible to micro-fracture under heavy loading. This histological reality dictates the standard unloaded healing period of 3 to 4 months in the mandible and 4 to 6 months in the maxilla within the dental implant process timeline.
Phase III: Remodeling and Lamellar Maturation
The final and longest phase of osseointegration is remodeling.
The Replacement of Woven Bone
Commencing around week 6 and continuing for years, the woven bone is gradually resorbed by osteoclasts and replaced by lamellar bone. Lamellar bone is organized into concentric layers (osteons) with collagen fibers aligned to resist functional stress. This process is governed by Wolff’s Law, which states that bone adapts to the loads under which it is placed. The timeframe for dental implants must account for this maturation. Premature loading before significant lamellar deposition can cause micro-damage accumulation at the interface, leading to retrograde peri-implantitis or loss of integration. The secondary stability achieved during this phase is permanent and increases over time as the bone mineral density improves.
Soft Tissue Dynamics and the Transmucosal Seal
Parallel to osseous healing, the soft tissue barrier must re-establish itself.
Hemidesmosomal Attachment
The peri-implant mucosa forms a biological seal around the transmucosal abutment. Unlike natural teeth, which have perpendicular connective tissue fibers inserting into the cementum, the fibers around an implant run parallel to the surface. The epithelial attachment is mediated by hemidesmosomes. The maturation of this soft tissue cuff takes approximately 4 to 8 weeks following second-stage surgery (uncovering). The stability of this seal is crucial for preventing bacterial ingress into the osseous zone. The dental implant process timeline includes this soft tissue healing phase to ensure the gingival architecture is stable prior to the final impression, preventing recession and aesthetic failure.
Variables Influencing the Timeline
The standard chronology is subject to numerous physiological variables.
Systemic and Local Modifiers
- Bone Density: Type IV bone (posterior maxilla) has larger marrow spaces and less cortical bone, requiring a longer timeframe for dental implants to achieve adequate bone-to-implant contact (BIC).
- Grafting Procedures: Sites requiring guided bone regeneration (GBR) or sinus augmentation introduce an additional variable. The graft material must first consolidate and be replaced by host bone (creeping substitution) before implant placement or loading can occur. This can extend the dental implant process timeline by 4 to 9 months.
- Systemic Health: Conditions such as uncontrolled diabetes or smoking impair microvascular circulation, delaying angiogenesis and extending the inflammatory phase.
The dental implant process timeline is a biological constant determined by cellular kinetics. The transition from a surgical wound to a functional load-bearing unit requires the undisturbed sequence of clotting, woven bone formation, and lamellar remodeling. Attempts to accelerate this timeframe for dental implants without respecting the histological limitations of the host tissue increase the risk of failure. Clinical success is achieved by synchronizing the restorative protocol with the patient’s specific healing velocity.